Research Hölzel Lab
Decoding Tumor Immunology
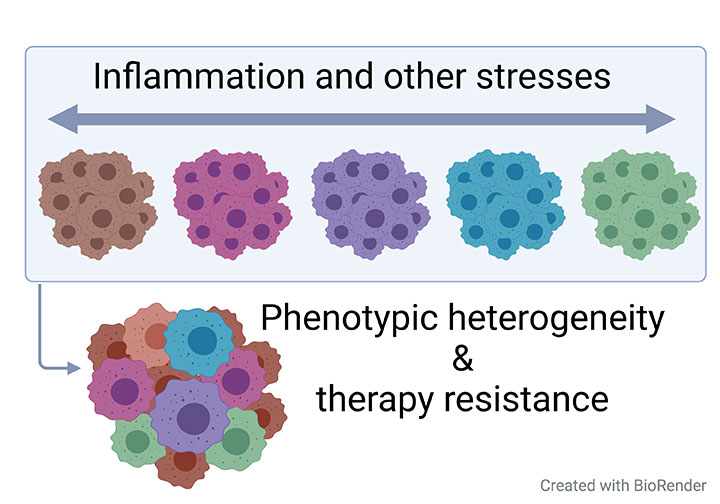
Tumor cell plasticity, inflammation and therapy resistance
We have a holistic view on the tumor tissue, as it is composed of different cellular compartments including tumor, immune, endothelial and stromal cells. All these different cell types are in constant exchange with each other, which provides a mechanistic basis for the dynamic adaptive changes that drive disease progression and therapy resistance.Within this scenario, a phenomenon called tumor cell plasticity plays a central role and it describes how tumor cells can switch their phenotypes in order to resist microenvironmental or therapeutic stress conditions. Initially focusing on malignant melanoma, an aggressive skin cancer originating from the pigment producing melanocytes, we explored how inflammatory signals from the tumor microenvironment act as key drivers of melanoma cell plasticity.
Currently, we are also studying the dynamic interplay between lipid metabolism, immune signaling and melanoma phenotype switching. Also, we have extended our view on other cancer types inspired by our many clinical collaborations, most notably urologic oncology, neuro-oncology and gynecology.
In our projects we aim to understand the molecular mechanisms that orchestrate tumor cell plasticity and identify drug targets to promote favorable rewiring of tumor cell phenotypes to overcome therapy resistance. For this, we are using a broad range of experimental approaches such as cultures of tumor cell lines and patient-derived organoids, preclinical in vivo models, various flavors of in vitro genetic engineering as well as multiparametric and high-dimensional analytical methods like flow cytometry, multiplex-IF (CODEX(R)) and genomics (scRNA-seq, RNA-seq, WES, WGS).
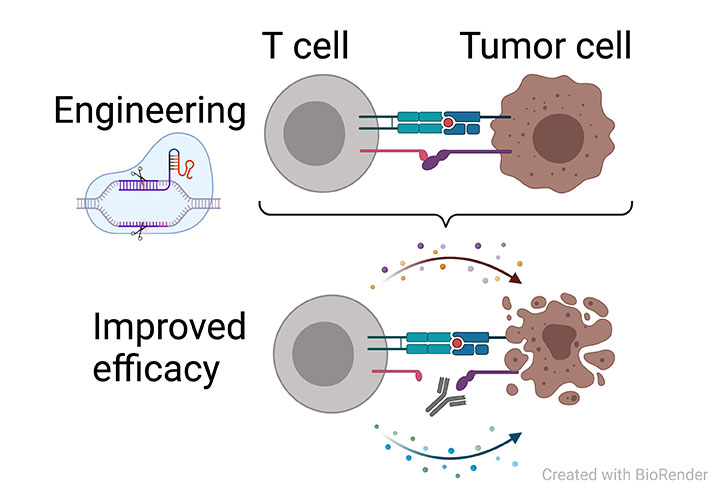
Exploring novel principles of adoptive cell transfer therapy
Adoptive cell transfer (ACT) is an emerging modality for personalized cancer immunotherapy. In brief, tumor-reactive T or NK cells, either isolated from tumors or genetically engineered, are re-infused in order to mount a potent anti-tumor immune response. Nevertheless, there are many unresolved aspects related to efficacy, durability and toxicity.We have developed a conceptional experimental platform termed CRISPR-assisted insertion of epitopes (CRISPitope) that allows us to study novel principle of adoptive T cell therapy in a standardized preclinical in vivo model.
By the means of CRISPR-Cas9 gene editing, we fuse model T cell epitopes to endogenous gene products, which then allows us to study and monitor how transferred epitope-specific T cells expand and contract, attack tumor cells and either succeed or fail in controlling tumor growth over time.
Using this versatile approach, we also aim to evaluate the development of common side effects of ACT by a systemic view. Furthermore, we established several innovative strategies how to improve current approaches of ACT and we will continue our path to work on novel clinically translatable treatment concepts.
Exploring novel principles of adoptive cell transfer therapy
Adoptive cell transfer (ACT) is an emerging modality for personalized cancer immunotherapy. In brief, tumor-reactive T or NK cells, either isolated from tumors or genetically engineered, are re-infused in order to mount a potent anti-tumor immune response. Nevertheless, there are many unresolved aspects related to efficacy, durability and toxicity.We have developed a conceptional experimental platform termed CRISPR-assisted insertion of epitopes (CRISPitope) that allows us to study novel principle of adoptive T cell therapy in a standardized preclinical in vivo model.
By the means of CRISPR-Cas9 gene editing, we fuse model T cell epitopes to endogenous gene products, which then allows us to study and monitor how transferred epitope-specific T cells expand and contract, attack tumor cells and either succeed or fail in controlling tumor growth over time.
Using this versatile approach, we also aim to evaluate the development of common side effects of ACT by a systemic view. Furthermore, we established several innovative strategies how to improve current approaches of ACT and we will continue our path to work on novel clinically translatable treatment concepts.
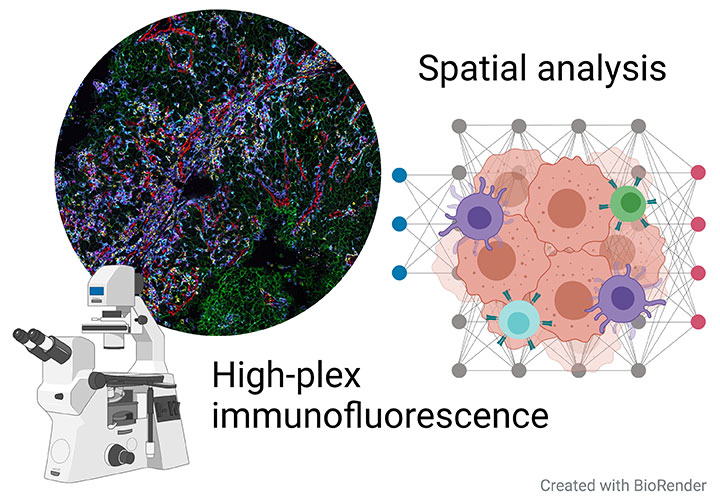
Deep spatial phenotyping of the human tumor microenvironment
In the recent years, several technologies have advanced rapidly that allow for a comprehensive analysis of tumor tissues on the single cell level, in particular single-cell RNA-sequencing (scRNA-seq) and multiparametric flow cytometry.High-plex immunofluorescence (high-plex IF) is another very recent method that allows to detect many different markers (>50) on a single tumor tissue section. By this, different cell types and their phenotypes can be analyzed while preserving the information about the spatial context of each cell, which is in contrast to scRNA-seq or flow cytometry.
High-plex IF therefore provides entirely novel insights into the complex architecture of tumor tissues and how different cell populations influence each other in spatial vicinity.
Hence, scRNA-seq, flow cytometry and high-plex IF are three complementary technologies and in conjunction allow us to analyze the human tumor microenvironment at so far unprecedented depth.
Having established the technical and analytical expertise, we aim to integrate the findings obtained from spatial deep phenotyping of human tumors with different types of clinical information, most importantly treatment responses to immune checkpoint inhibitors or latest antibody drug conjugates. By this, we expect to learn more about the heterogeneity of treatment responses seen in patients and delineate novel counterstrategies to overcome resistance.
Our deep phenotyping approaches are complemented by a concerted effort with our clinical collaborators from University Hospital Bonn (UKB) employing patient-derived organoids and short-term cultures of tumor fragments to investigate drug responses.
Join the team and explore more novel principles
Interested in more information about our projects?
Further Reading
Druggable epigenetic suppression of interferon-induced chemokine expression linked to MYCN amplification in neuroblastoma.
Seier JA, Reinhardt J, Saraf K, Ng SS, Layer JP, Corvino D, Althoff K, Giordano FA, Schramm A, Fischer M, Hölzel M
J Immunother Cancer. 2021;9(5). doi:10.1136/jitc-2020-001335
J Immunother Cancer. 2021;9(5). doi:10.1136/jitc-2020-001335
Adoptive T Cell Therapy Targeting Different Gene Products Reveals Diverse and Context-Dependent Immune Evasion in Melanoma.
Effern M, Glodde N, Braun M, Liebing J, Boll HN, Yong M, Bawden E, Hinze D, van den Boorn-Konijnenberg D, Daoud M, Aymans P, Landsberg J, Smyth MJ, Flatz L, Tüting T, Bald T, Gebhardt T, Hölzel M
Immunity. 2020;53(3):564-580.e9.
Immunity. 2020;53(3):564-580.e9.
Reactive Neutrophil Responses Dependent on the Receptor Tyrosine Kinase c-MET Limit Cancer Immunotherapy.
Glodde N, Bald T, van den Boorn-Konijnenberg D, Nakamura K, O’Donnell JS, Szczepanski S, Brandes M, Eickhoff S, Das I, Shridhar N, Hinze D, Rogava M, van der Sluis TC, Ruotsalainen JJ, Gaffal E, Landsberg J, Ludwig KU, Wilhelm C, Riek-Burchardt M, Müller AJ, Gebhardt C, Scolyer RA, Long GV, Janzen V, Teng MWL, Kastenmüller W, Mazzone M, Smyth MJ, Tüting T, Hölzel M
Immunity. 2017;47(4):789-802.e9.
Immunity. 2017;47(4):789-802.e9.
MAPK Signaling and Inflammation Link Melanoma Phenotype Switching to Induction of CD73 during Immunotherapy.
Reinhardt J, Landsberg J, Schmid-Burgk JL, Ramis BB, Bald T, Glodde N, Lopez-Ramos D, Young A, Ngiow SF, Nettersheim D, Schorle H, Quast T, Kolanus W, Schadendorf D, Long GV, Madore J, Scolyer RA, Ribas A, Smyth MJ, Tumeh PC, Tüting T, Hölzel M
Cancer Res. 2017;77(17):4697–4709.
Cancer Res. 2017;77(17):4697–4709.
MITF and c-Jun antagonism interconnects melanoma dedifferentiation with pro-inflammatory cytokine responsiveness and myeloid cell recruitment.
Riesenberg S, Groetchen A, Siddaway R, Bald T, Reinhardt J, Smorra D, Kohlmeyer J, Renn M, Phung B, Aymans P, Schmidt T, Hornung V, Davidson I, Goding CR, Jönsson G, Landsberg J, Tüting T, Hölzel M
Nature Commun. 2015;6:8755.
Nature Commun. 2015;6:8755.